Reversible Denaturation of Proteins
Background
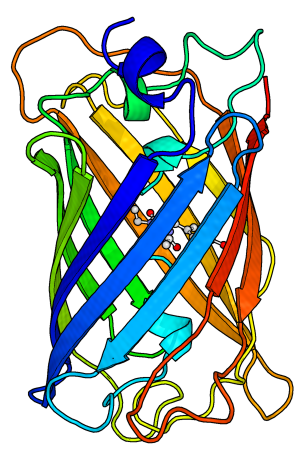
Some proteins can be reversibly folded and unfolded under various conditions. In this experiment we will use acid to denature proteins, and then reverse this by neutralizing the denatured protein solution with the addition of base.
To observe the effect of a small amount of a denaturing chemical on a protein’s structure, we will use green fluorescent protein, which emits green light when properly folded (so the sample looks green in color).
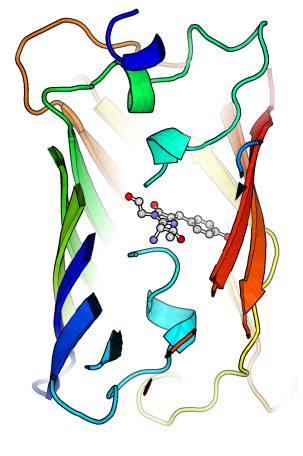
This is a useful readout because the green color will disappear when the green fluorescent protein is denatured, as a result of having the chromophore (substance that produces the fluorescent green light) no longer protected by the surrounding protein structure. The addition of base will neutralize the acid and allow this very resilient protein to refold around the chromophore and regain its green color.
Materials

Corner Store Items
- Egg (ideally separated; see below)
- Strainer

Lab Equipment
- Microfuge Tubes or Test Tubes
- Micropipette and tips or Pasteur pipette with a rubber bulb

Lab Items
- GFP (green fluorescent protein)*
- 0.5 M HCl
- 0.5 M NaOH
*GFP can be purchased from some chemical supply companies, expressed and purified yourself or obtained from a laboratory with leftover samples (as was the motivation for this experiment in the first place). If access to GFP is challenging, use the accompanying video to observe this process virtually.
Preparation (optional but recommended)

In this raw egg, observe the tight white (thick and well defined around the yolk) and the loose white (spreading thinly along the table).

Put the egg white into a fine mesh strainer that is situated over a beaker or cup. This will separate the egg white into the tight white (close to the yolk, thicker due to less water content; held together by a very thin membrane) and the loose white (the outermost egg white, thinner due to more water content).
Allow the egg white sit to slowly pass through the strainer, do not agitate significantly. The tight white will remain in the strainer and the loose white will drip into the beaker or cup.
Procedure
- Begin with 100 µL aliquots of GFP, loose egg white, tight egg white, and egg yolk in separate clear microfuge tubes/test tubes.
- Add 3 µl (or a single drop from a Pasteur pipette) 0.5 M HCl to each sample and observe.
What conclusions can you draw about how the acid affects the GFP and egg protein structure? Describe the changes you observe in your samples.
- To neutralize each sample you will need to add 3 µl 0.5 M NaOH to start. You will then have to add more base, in 1 µl drops, being sure to swirl gently between additions to get any acid off the side of the tube and make sure the sample is evenly mixed. (Alternatively, add base in single drops from a Pasteur pipette).
What conclusions can you draw about how the base affects the GFP and egg protein structure? Describe the changes you observe in your samples.
- You may experiment with adding more (or stronger) acid as well, but the GFP structure will not refold under conditions that are too extreme.
Why might the GFP structure not refold under extreme conditions?
Virtual GFP Demo
Green fluorescent protein (GFP) protein is likely hard to come by. Check out this video to see the demonstration and compare it to your hands-on exploration of the proteins in egg.
Discussion
Why is the primary structure of proteins unaffected by denaturation?
Do all proteins denature under the same conditions? Why or why not? (specify under same temp, pH, etc.)
Identify some possible factors that can cause protein denaturation.Are proteins denatured in the human stomach? Why or why not?