Relate to Emulsions
Read about some emulsions that play important roles in our everyday lives through these Case Study readings and reflection questions.
Deepen your knowledge...
Case Study: Lipid Emulsions and Parenteral Nutrition
Background
Macronutrients such as carbohydrates, protein, and fat (lipid), are essential components of the human diet. Under normal circumstances, people are able to obtain the majority of these macronutrients by eating healthy foods. Our bodies ultimately absorb macronutrients from food in the small intestine, but not before our digestive processes prepare these macronutrients into absorbable compounds. For example, ingested dietary fat cannot just enter the small intestine — it is first acted on in the mouth, where mechanical and enzymatic processes begin to emulsify dietary fat, which is further acted upon in the stomach, also by enzymes and mechanical processes.
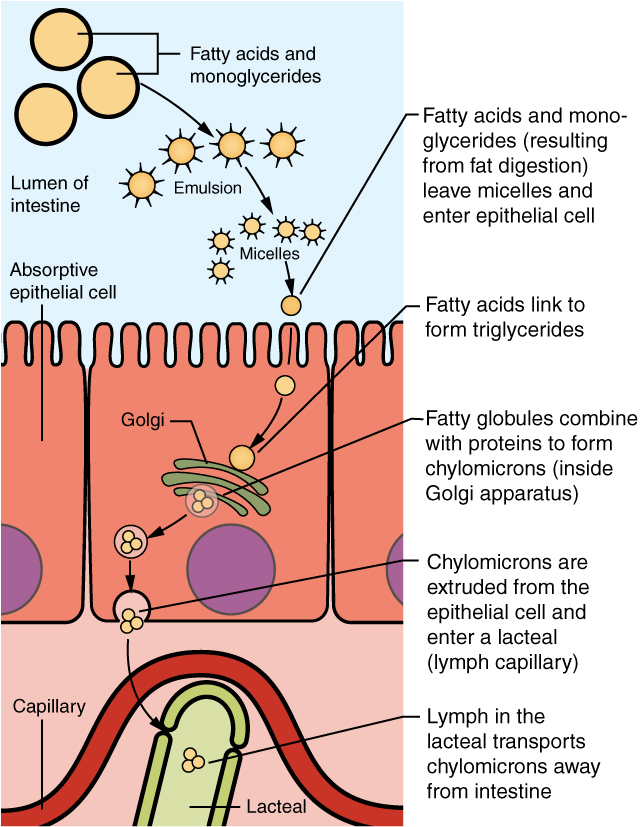
Figure 1: Lipid absorption process (source: wikimedia)
Once stomach contents are transferred to the small intestine, these partially emulsified dietary fats are exposed to bile, which finishes the emulsification process. Fully emulsified fat particles can now pass into the cells lining the small intestine, where they are packed up into tiny spheres of protein and fat called chylomicrons.
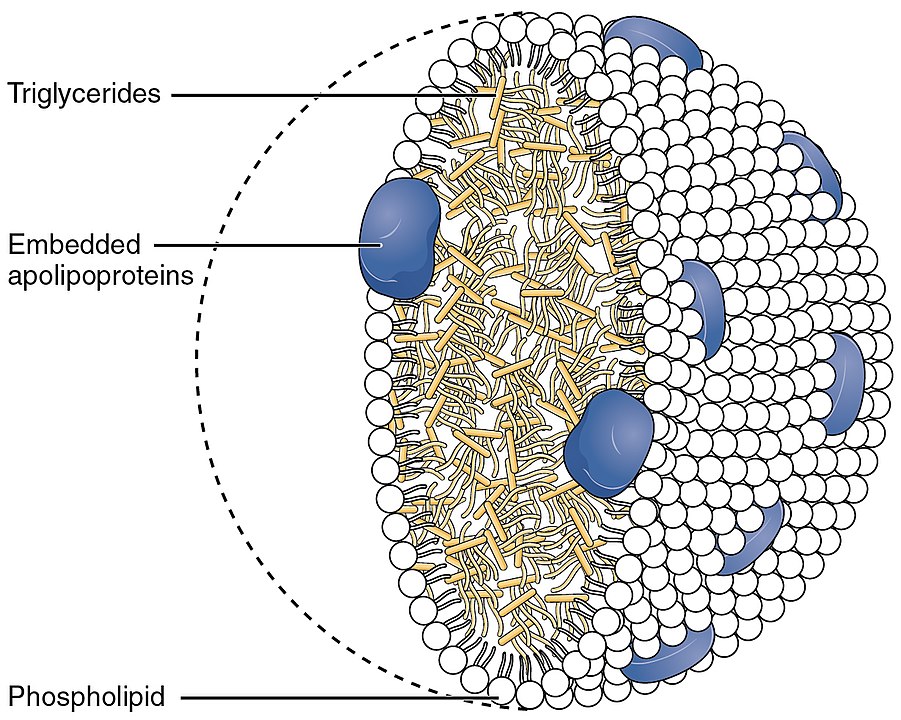
Figure 2: Chylomicron diagram (source: wikimedia)
From there, chylomicrons enter the bloodstream, allowing for the delivery of lipids to our tissues.
What happens when someone experiences issues with digestion and nutrient absorption?
There are instances of diseases or disorders related to impaired gut function that make it difficult or impossible for people to obtain adequate nutrition. When a patient is challenged by these issues, a common treatment is to supplement them through intravenous administration of nutrients, otherwise known as parenteral nutrition (PN). A major component of PN mixtures include Intravenous Lipid Emulsions (ILE), which provide patients with a source of essential fatty acids and non-protein calories. However, long-term use of ILEs has been associated with intestinal complications and liver disease. As such, scientists and clinicians have been working to engineer a better recipe for ILEs that can provide needed nutrition without harm.
Note: because PN is delivered through an IV, this mode of feeding bypasses the normal digestion processes that take place in the gastrointestinal tract. Instead, the ILEs contained in the PN are absorbed and processed by the small intestine and liver.
Case Study: Emulsions to Keep Clean
Review & Reflect
1. For each example, what kinds of molecules are you trying to clean away? Dishes • Hair • Hands • Clothes
2. Considering #1, why might you need separate dish soap, hand soap, laundry detergent and shampoo?
3. The verb to shampoo comes from Hindi chāmpo (चाँपो [tʃãːpoː]), itself derived from the Sanskrit root chapati (चपति), which means to press, knead, soothe. How does this relate to the emulsion process? Could these early meanings apply to any of the cleaning categories or just to shampoo?
Now learn more by reading at least one of the following:
Case Study: Lipid Nanoparticles and mRNA Vaccine Delivery
Background
Existing at the crossroads of immunology and molecular biology, the mRNA vaccine against SARS-CoV-2, the virus that causes COVID-19, is the ultimate example of how decades of basic biomedical research can culminate as a global response to an infectious disease. It might be obvious that the “mRNA” part of the “mRNA vaccine” is what is technically responsible for building our immunity, but likely less appreciated is how the mRNA is able to get into our cells in the first place. The US-based pharmaceutical companies, Moderna and Pfizer-BioNTech, both created a vaccine that delivers specific mRNA molecules into our cells using lipid biochemistry, specifically focusing on emulsions. Representing a medical application of emulsion science, Lipid Nanoparticles (LNPs) are designed as a highly specialized transport system for protecting mRNA molecules. These tiny and highly specialized lipid spheres get vaccine-specific mRNA into our cells so they do their immunological thing.
mRNA-LNP Structure
mRNA-LNPs have been called a “true platform technology.” Once scientists understood the rules of how cellular molecules such as lipids, proteins, and nucleotides can interact, the creation of LNPs for mRNA vaccine delivery became streamlined, translating into a relatively quick turnaround for specific mRNA vaccines. LNPs contain 4 major lipid molecules, each serving important purposes (Table 1):
Table 1: LNP lipid components and their function
| LNP Lipid | Purpose |
| Helper Phospholipid | Structural elements of LNPs that dictate LNP membrane architecture and biocompatibility, including characteristics like size, curvature, fluidity, and charge. PEG Lipids specifically help prevent particle aggregation during storage |
| Cholesterol | |
| Polyethylene Glycol (PEG) Lipid | |
| Ionizable Cationic Lipid | A lipid that is neutral as it travels through our bloodstream, but can switch to a positive charge when specific conditions are met |
To make the COVID-19 mRNA vaccine, the above lipid components are rigorously combined in specific ratios and at a specific rate with modified mRNA strands that map to the spike (S) protein of the SARS-CoV-2 virus. This process ultimately creates densely packed LNPs, with mRNA molecules safely encapsulated in lipids. While the precise architecture of mRNA-LNPs remains unknown (for now), the general principle is represented in Figure 1. Essentially, Ionizable Cationic Lipids surround the negatively charged mRNA molecules. These lipid-mRNA complexes are further encapsulated by a mixed-lipid coat (PEG-Lipid, Ionizable Cationic Lipid, Helper Lipid, and Cholesterol). This particle is taken up by cells, and the mRNA is released into the cytosol where it begins the process of immunological programming.

Figure 1: Schematic of an mRNA-LNP (Illustration by Marc Roseboro/CNSI)
mRNA-LNP Action
Recall when you got your COVID-19 vaccine or flu shot — the needle went right into your upper arm. This is called Intramuscular (IM) administration, and most vaccines are delivered into our bodies via this route. The IM injection itself causes localized inflammation and the recruitment of immune cells to the injection site. The mRNA-LNPs fuse with the cell membranes of these immune system cellular recruits, ultimately dumping the mRNA into the cytosol, where it can be translated into the S protein. The S proteins are then presented on the surface of these cells as an “antigen,” setting off the process to build up immunity against COVID-19.
Case Study: Palmitic Acid
Background
The most abundant fatty acid in nearly all organisms on earth is palmitic acid (C16) — a saturated fatty acid with 16 carbons. The main food sources of C16 include meat, animal fats, milk, dairy products, eggs, and vegetable oils – mainly palm oil. C16 in our body serves as the building block of many other fatty acids, including but not limited to phospholipids, the fundamental component of cell membranes.
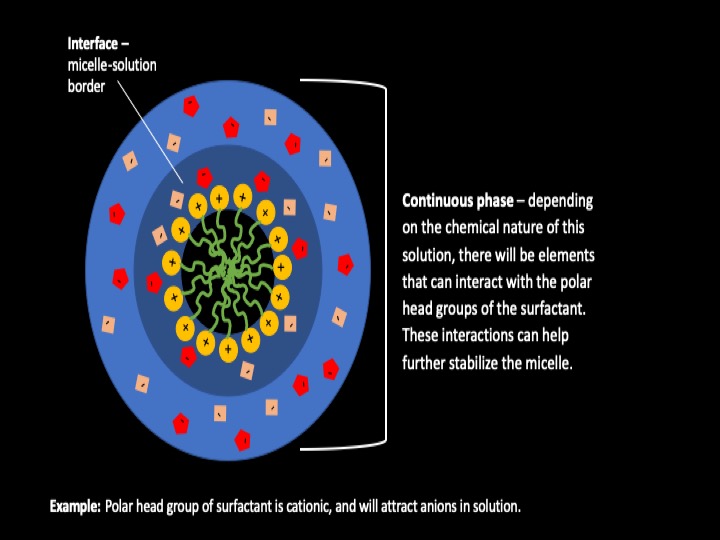
Emulsion diagram
In our bodies, C16 content of each tissue is tightly regulated, and under normal physiological conditions, does not readily deviate. This strict homeostatic maintenance of C16 content in tissues is likely a function of the fundamental role C16 plays in many biological processes. Not only does C16 give rise to fuel sources that create energy and heat in the body, it also stabilizes cell membranes, protects nerve and brain cells, and acts as cell-signaling carriers. C16 also acts as surfactants in our lungs that reduces surface tension in our alveoli to prevent collapse when exhaling!
As much as the emulsifier quality of C16 serves many essential functions in our body, it also induces some negative effects that can possibly become mortally dangerous. Over- accumulation of C16 caused by high consumption of carbohydrates and sedentary lifestyle elevates levels of emulsified LDL (Bad) cholesterol in the blood that can give rise to heart disease as well as inflammation. The destabilizing effect of surfactants can also structurally disorganize membranes and denature proteins including essential enzymes related to DNA repair, aiding the proliferation of cancer cells.
Emulsifying quality of C16 can have two totally opposite effects depending on the concentration and other biological, physiological causes. C16 is a telling evidence that biology is heavily dependent on homeostasis, or finding the optimal balance between components.