Case Study: Palmitic Acid
Image source: https://www.scienceofcooking.com/palmitic_acid.htm
Background
The most abundant fatty acid in nearly all organisms on earth is palmitic acid (C16) — a saturated fatty acid with 16 carbons. The main food sources of C16 include meat, animal fats, milk, dairy products, eggs, and vegetable oils – mainly palm oil. C16 in our body serves as the building block of many other fatty acids, including but not limited to phospholipids, the fundamental component of cell membranes.
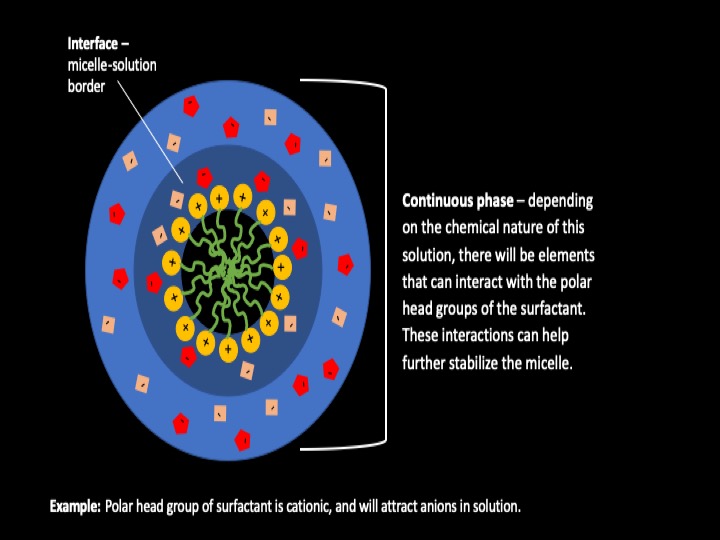
Emulsion diagram
In our bodies, C16 content of each tissue is tightly regulated, and under normal physiological conditions, does not readily deviate. This strict homeostatic maintenance of C16 content in tissues is likely a function of the fundamental role C16 plays in many biological processes. Not only does C16 give rise to fuel sources that create energy and heat in the body, it also stabilizes cell membranes, protects nerve and brain cells, and acts as cell-signaling carriers. C16 also acts as surfactants in our lungs that reduces surface tension in our alveoli to prevent collapse when exhaling!
As much as the emulsifier quality of C16 serves many essential functions in our body, it also induces some negative effects that can possibly become mortally dangerous. Over- accumulation of C16 caused by high consumption of carbohydrates and sedentary lifestyle elevates levels of emulsified LDL (Bad) cholesterol in the blood that can give rise to heart disease as well as inflammation. The destabilizing effect of surfactants can also structurally disorganize membranes and denature proteins including essential enzymes related to DNA repair, aiding the proliferation of cancer cells.
Emulsifying quality of C16 can have two totally opposite effects depending on the concentration and other biological, physiological causes. C16 is a telling evidence that biology is heavily dependent on homeostasis, or finding the optimal balance between components.
Reflect & Relate
1. What are the potential implications (both positive and negative) of emulsifiers in biological setting?
2. What are the mechanisms of C16 induction of cancer or inflammation?
3. What molecules can surfactants destabilize and what can it not? Is there a limit to emulsifiers’ penetrating ability?
4. What are some other biological, chemical contexts in which homeostasis is essential?
5. How can this relate to classwork in your setting?









